热烈祝贺博士研究生张迪和牟方志研究员等撰写的研究论文“Systemic-to-Local Nanorobot Thrombolysis”被Science子刊Science Advances(2025年IF: 12.5,中科院 Q1期刊)接收!
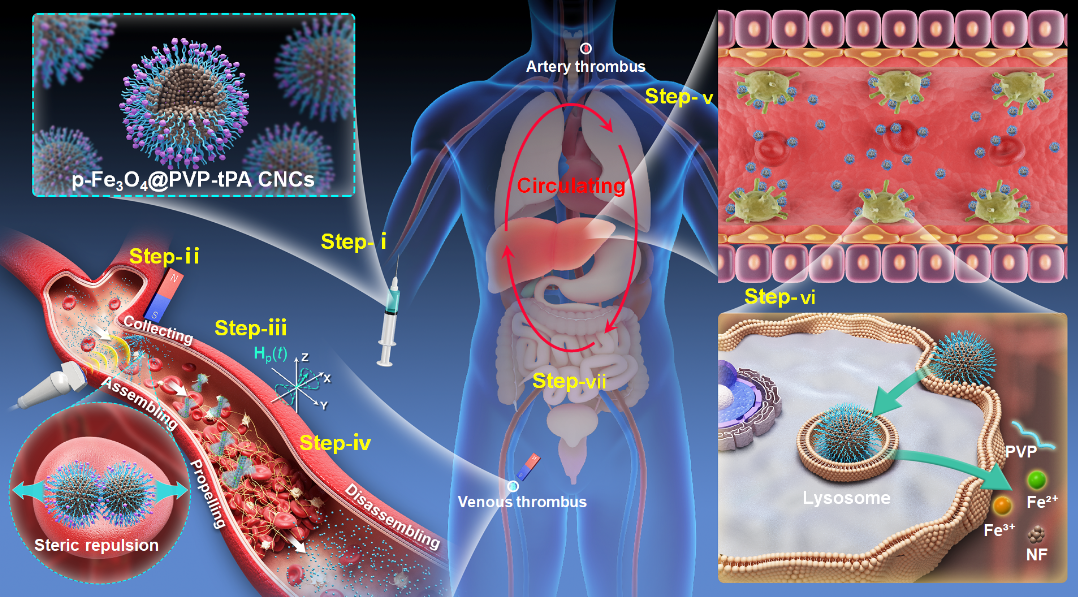
磁性微/纳米机器人在靶向溶栓治疗中展现出巨大潜力,但仍面临快速免疫清除和代谢降解性差等挑战,往往只能实施侵入式的局部给药并需要回收。本文提出了一种安全且通用的系统给药-局部溶栓策略,其核心是用聚乙烯吡咯烷酮(PVP)屏蔽的多孔Fe₃O₄ 胶体纳米晶簇(p-Fe₃O₄@PVP CNCs)构建磁性纳米机器人。该设计精细平衡了长循环稳定性、强磁响应性和快速生物降解性。其中,PVP壳层不仅能负载组织型纤溶酶原激活剂(tPA),而且能确保CNCs经静脉系统给药后在血液中实现长循环。CNCs的内核则提供强的集体磁矩,确保CNCs能通过梯度磁场进行磁收集,再在进动磁场驱动下原位形成可导航的磁驱纳米机器人,执行靶向溶栓。在溶栓治疗完成撤去磁场后,纳米机器人自动解离为分散的CNCs。由于CNCs为超小初级纳米晶(<5 nm)组成的多孔结构,它能够在溶酶体中快速降解,并主要通过肝-胆-肠轴排出体外,从而能避免长期毒性。该平台为纳米机器人溶栓治疗的临床转化提供了一种能克服关键瓶颈的解决方案。
原文摘要如下:Magnetic micro/nanorobots hold promise for targeted thrombolysis, yet face challenges of rapid immune clearance and poor metabolic degradability, often necessitating invasive localized deployment and retrieval. Here, we present safe and versatile systemic-to-local thrombolysis enabled by magnetic nanorobots constructed from polyvinylpyrrolidone-shielded porous Fe3O4 colloidal nanocrystal clusters (p-Fe3O4@PVP CNCs). This design elaborately harmonizes prolonged circulation, strong magnetic responsiveness, and rapid biodegradability. The stealth PVP coating allows tPA-loaded CNCs to be systemically injected, achieving prolonged blood persistence. The p-Fe3O4 nanocrystal-cluster core provides a strong collective magnetic moment necessary for sequential magnetic collection (via gradient field H) and actuation into navigating nanorobots (via precessing field Hp(t)) for targeted thrombolysis. After therapy, the disassembled CNCs, owing to their porous structure and ultrasmall primary nanocrystals (<5 nm), undergo rapid lysosomal degradation and are cleared primarily via the liver–bile–intestine axis, resulting in no long-term toxicity. This platform overcomes key translational challenges for nanorobotic thrombolytic therapy.